GROUP 17 ELEMENTS
- The elements in Group 17 are also known as the halogens.
- The elements in Group 17 are flourine (F), chlorine (Cl), bromine (Br), iodine (I) and astatine (At).
- Halogens are very reactive elements and most of them exist naturally as halide salts.
- The halogen molecules exists as diatomic molecules: F2, Cl2, Br2, I2, and At2
PHYSICAL PROPERTIES OF HALOGENS
- All Group 17 elements are non-metals. Hence, they are insulators of heat and electricity.
- The atomic radius increase down the group. The reason is that as the number of filled electron shell increase down the group, the distance between the outermost electron shell and the nucleus increase.
- The density increases down the group. This is due to the increase in relative molecular mass.
- Halogens have low boiling points. The forces of attraction between the molecules are weak.
- The melting points and boiling points of the halogens increase down the group. This is because the molecular size increases down the group.
- As the size increases, the van der Waal`s forces of attraction between the molecules become stronger. More heat is required to overcome the attractive forces and therefore the melting points and boiling points increase. The first two elements (fluorine and chlorine) are gases at room temperature. Bromine is a liquid whereas iodine and astatine are solids at room temperature.
- The colour of the halogen becomes darker down the group. Fluorine is a colourless gas; chlorine is a yellowish green gas; bromine is a dark brown liquid and iodine is a black solid.
- All halogens have high electro-negativities. They are electronegative non-metals. Electronegativity is a measurement of the element to attract electrons.
Some Physical Properties of Three Halogens
- The elements in Group 17 are flourine (F), chlorine (Cl), bromine (Br), iodine (I) and astatine (At).
- Halogens are very reactive elements and most of them exist naturally as halide salts.
- The halogen molecules exists as diatomic molecules: F2, Cl2, Br2, I2, and At2
PHYSICAL PROPERTIES OF HALOGENS
- All Group 17 elements are non-metals. Hence, they are insulators of heat and electricity.
- The atomic radius increase down the group. The reason is that as the number of filled electron shell increase down the group, the distance between the outermost electron shell and the nucleus increase.
- The density increases down the group. This is due to the increase in relative molecular mass.
- Halogens have low boiling points. The forces of attraction between the molecules are weak.
- The melting points and boiling points of the halogens increase down the group. This is because the molecular size increases down the group.
- As the size increases, the van der Waal`s forces of attraction between the molecules become stronger. More heat is required to overcome the attractive forces and therefore the melting points and boiling points increase. The first two elements (fluorine and chlorine) are gases at room temperature. Bromine is a liquid whereas iodine and astatine are solids at room temperature.
- The colour of the halogen becomes darker down the group. Fluorine is a colourless gas; chlorine is a yellowish green gas; bromine is a dark brown liquid and iodine is a black solid.
- All halogens have high electro-negativities. They are electronegative non-metals. Electronegativity is a measurement of the element to attract electrons.
Some Physical Properties of Three Halogens
REACTIVITY OF GROUP 17 ELEMENTS
- All Group 17 elements are very reactive. However, the reactivity decreases down the group.
- The chemical reactions of Group 17 elements involve the formation of negative ions of charge –1: Cl -, Br -, I-
- The elements in Group 17 have 7 valence electrons each. During a chemical reaction, the atom of a Group 17 element will gain a valence electron to form univalent negative ion to attain the stable octet in its electron arrangement.
- All Group 17 elements are very reactive. However, the reactivity decreases down the group.
- The chemical reactions of Group 17 elements involve the formation of negative ions of charge –1: Cl -, Br -, I-
- The elements in Group 17 have 7 valence electrons each. During a chemical reaction, the atom of a Group 17 element will gain a valence electron to form univalent negative ion to attain the stable octet in its electron arrangement.
- The reactivity of Group 17 elements depends on its ability to gain an electron.
- The atomic radius of Group 17 elements increases down the group. Thus the forces of attraction between the nucleus and the electrons become weaker.
- As the result, the halogen lower in the group has a lower tendency to attract an electron to form negative ion. Therefore, the reactivity of halogens decreases down the group.
CHEMICAL PROPERTIES
- The elements in Group 17 have the same chemical properties because each have seven valence electrons
- Group 17 elements react with:
(i) water to produce acids.
(ii) metals such as iron to produce metal halides.
(iii) sodium hydroxide to produce salts and water.
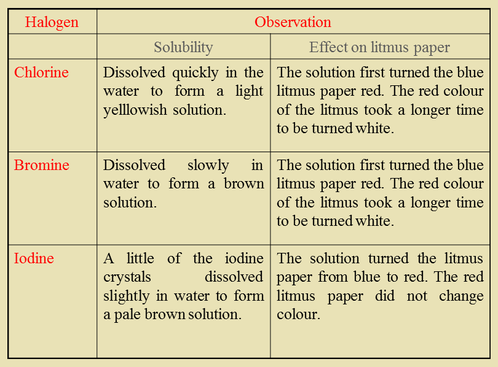
The Reaction of Chlorine, Bromine and Iodine with Water
- We notice that chlorine, bromine and iodine have similar chemical properties but differ in reactivity. Halogens react with water to form two acids. For example,
- The atomic radius of Group 17 elements increases down the group. Thus the forces of attraction between the nucleus and the electrons become weaker.
- As the result, the halogen lower in the group has a lower tendency to attract an electron to form negative ion. Therefore, the reactivity of halogens decreases down the group.
CHEMICAL PROPERTIES
- The elements in Group 17 have the same chemical properties because each have seven valence electrons
- Group 17 elements react with:
(i) water to produce acids.
(ii) metals such as iron to produce metal halides.
(iii) sodium hydroxide to produce salts and water.
The Reaction of Chlorine, Bromine and Iodine with Water
- We notice that chlorine, bromine and iodine have similar chemical properties but differ in reactivity. Halogens react with water to form two acids. For example,
The Reaction of Halogens with Aqueous Sodium hydroxide Solution
- Halogens also react with sodium hydroxide solution, NaOH to form sodium halate(I) and water. For example,
- Halogens also react with sodium hydroxide solution, NaOH to form sodium halate(I) and water. For example,
The Reactions of Halogens with Iron Wool
- Halogens in gaseous state react with hot iron to form a brown solid, iron (III) halides. For example,
- Halogens in gaseous state react with hot iron to form a brown solid, iron (III) halides. For example,
SAFETY PRECAUTIONS
- Fluorine is a very dangerously reactive substance whereas astatine is radioactive.
- Fluorine gas, chlorine gas, bromine vapour and iodine vapour are poisonous.
- Must handle them in a fume chamber. Besides that, must also wear safety goggles and gloves when handling these halogens.
- Fluorine is a very dangerously reactive substance whereas astatine is radioactive.
- Fluorine gas, chlorine gas, bromine vapour and iodine vapour are poisonous.
- Must handle them in a fume chamber. Besides that, must also wear safety goggles and gloves when handling these halogens.