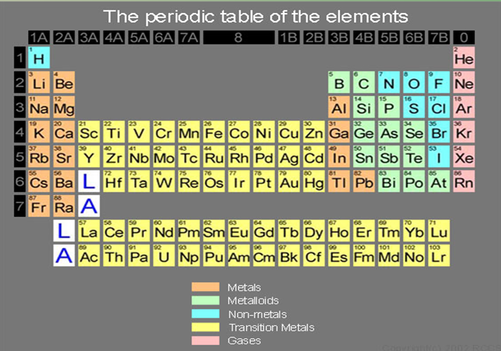
Arrangement Of Element In The Period Table
- Elements are arranged horizontally in ascending order of their proton numbers in the periodic table.
- Each vertical column of elements in the periodic table is known as a group. Elements with the same number of valence electrons are arranged in the same group.
- There are 18 vertical columns of elements in the periodic table, known as Group 1, Group 2, Group 3 until Group 18.
- Group 1 elements are known as alkali metals. Group 2 elements are known as alkali earth metals. Group 3 to Group 12 elements are known as transition elements. Group 17 elements are known as halogens. Group 18 elements are known as noble gases.
- Each horizontal row of elements in the periodic table is known as a period.
- There are 7 horizontal rows in the periodic table, known as Period 1, Period 2, Period 3 until Period 7.
- Period 1 – 2 elements,
- Periods 2 & 3 – 8 elements respectively.
- Periods 4 & 5 – 18 elements each.
- Periods 6 – 32 elements.
- Period 7 – 23 elements.
- Periods 1 to 3 are short periods while Periods 4 to 7 are long periods.
- Although Period 6 contains 32 elements, elements with proton numbers 58 to 71 are listed separately at the bottom of the periodic table. This series of elements is known as lantanides.
- Similarly, elements with proton numbers 90 to 103 in Period 7 are listed separately at the bottom of the Periodic Table. This series of elements is known as actinides.
- Elements in Groups 1, 2 and 13 are metals.
- Transition elements in Groups 3 to 12 are also metals.
- Non-metals usually have 5, 6 or 7 valence electrons. They are placed in Groups 15, 16 and 17 respectively in the Periodic Table. Carbon and silicon from Group 14 are also non-metals.
- Each vertical column of elements in the periodic table is known as a group. Elements with the same number of valence electrons are arranged in the same group.
- There are 18 vertical columns of elements in the periodic table, known as Group 1, Group 2, Group 3 until Group 18.
- Group 1 elements are known as alkali metals. Group 2 elements are known as alkali earth metals. Group 3 to Group 12 elements are known as transition elements. Group 17 elements are known as halogens. Group 18 elements are known as noble gases.
- Each horizontal row of elements in the periodic table is known as a period.
- There are 7 horizontal rows in the periodic table, known as Period 1, Period 2, Period 3 until Period 7.
- Period 1 – 2 elements,
- Periods 2 & 3 – 8 elements respectively.
- Periods 4 & 5 – 18 elements each.
- Periods 6 – 32 elements.
- Period 7 – 23 elements.
- Periods 1 to 3 are short periods while Periods 4 to 7 are long periods.
- Although Period 6 contains 32 elements, elements with proton numbers 58 to 71 are listed separately at the bottom of the periodic table. This series of elements is known as lantanides.
- Similarly, elements with proton numbers 90 to 103 in Period 7 are listed separately at the bottom of the Periodic Table. This series of elements is known as actinides.
- Elements in Groups 1, 2 and 13 are metals.
- Transition elements in Groups 3 to 12 are also metals.
- Non-metals usually have 5, 6 or 7 valence electrons. They are placed in Groups 15, 16 and 17 respectively in the Periodic Table. Carbon and silicon from Group 14 are also non-metals.
For elements with 1 or 2 valence electrons, the group number of that element is equal to the number of valence electrons.
For elements with 3 to 8 valence electrons, the group number of that element is equal to the number of valence electrons plus 10.
For elements with 3 to 8 valence electrons, the group number of that element is equal to the number of valence electrons plus 10.
- The numbers of shells occupied with electrons in an atom determines the position of the period of that element in the Periodic Table.
- Hence, the position of the period of an element is equal to the number of shells occupied with electrons in the atom of that element.
Advantages Of Classifying Elements in the Periodic Table
- Enables chemist to learn and understand physical properties and chemical properties of the elements and compounds more systematically, orderly and easily.
- The properties of an element and its compounds can be predicted based on its position in the Periodic Table.
- It becomes easier to study and understand the relationship among the elements from different groups.
- Enables chemist to learn and understand physical properties and chemical properties of the elements and compounds more systematically, orderly and easily.
- The properties of an element and its compounds can be predicted based on its position in the Periodic Table.
- It becomes easier to study and understand the relationship among the elements from different groups.